For Professionals
For Professionals For Professionals
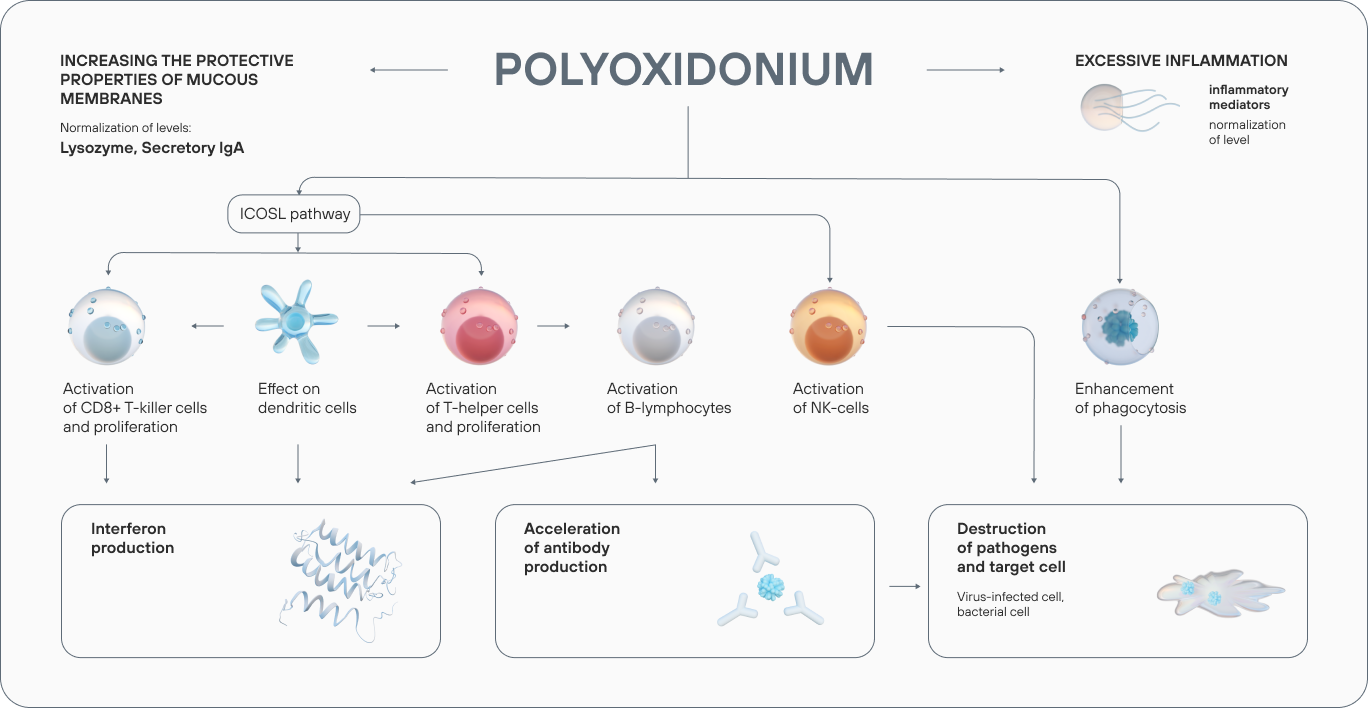
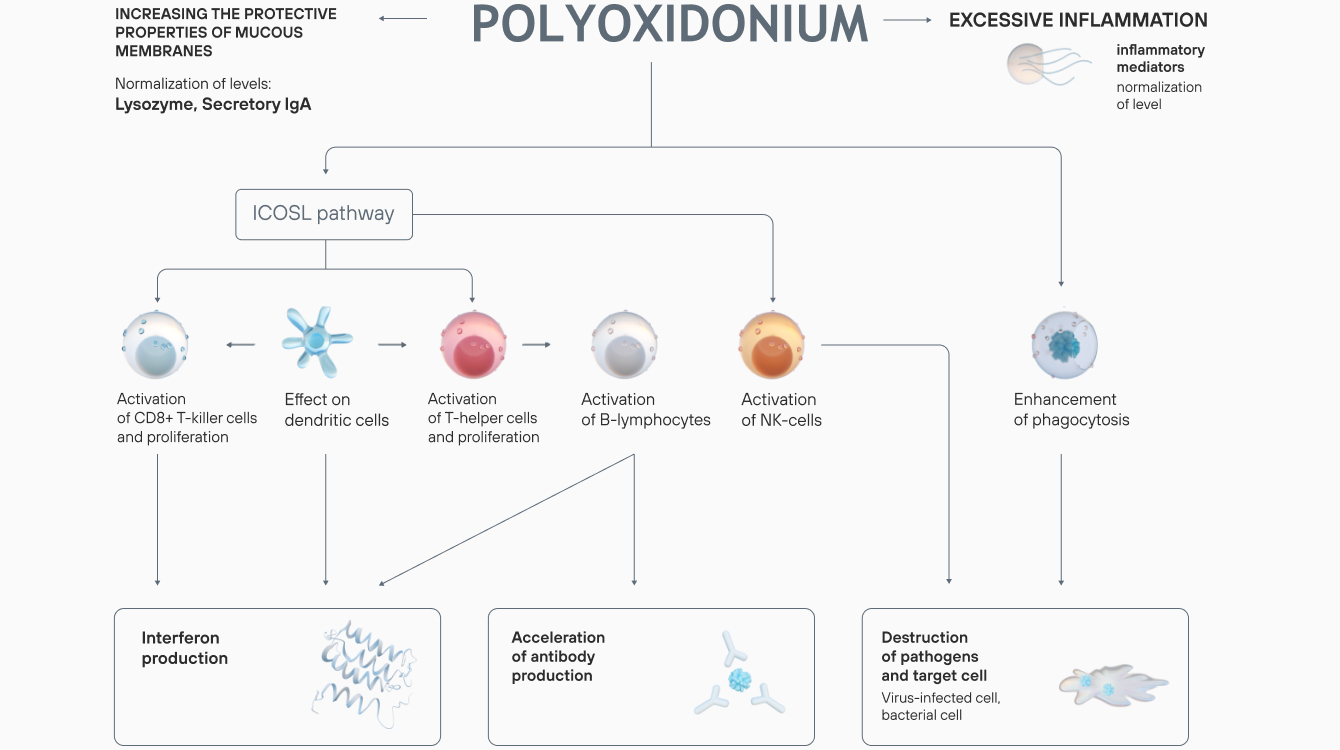
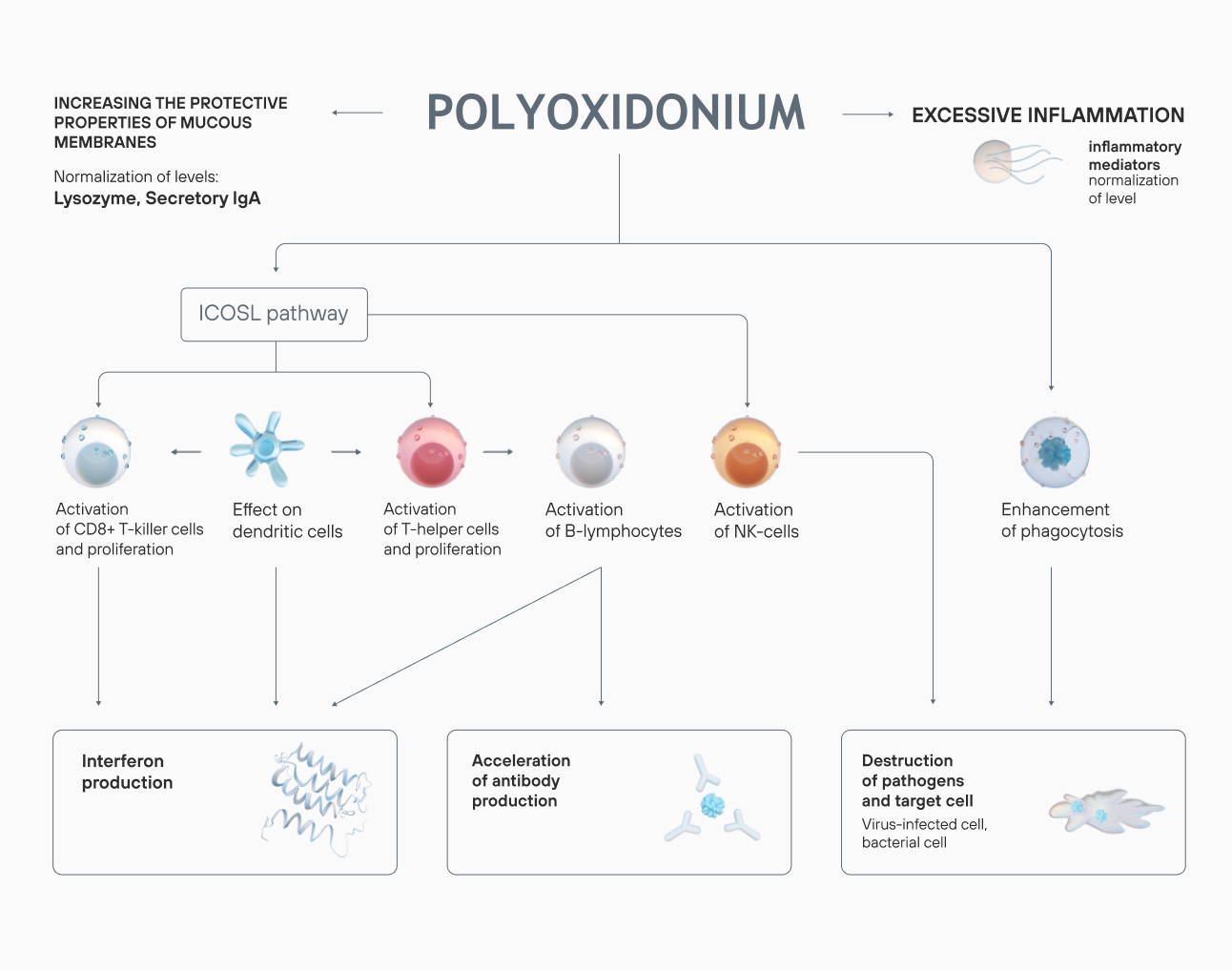
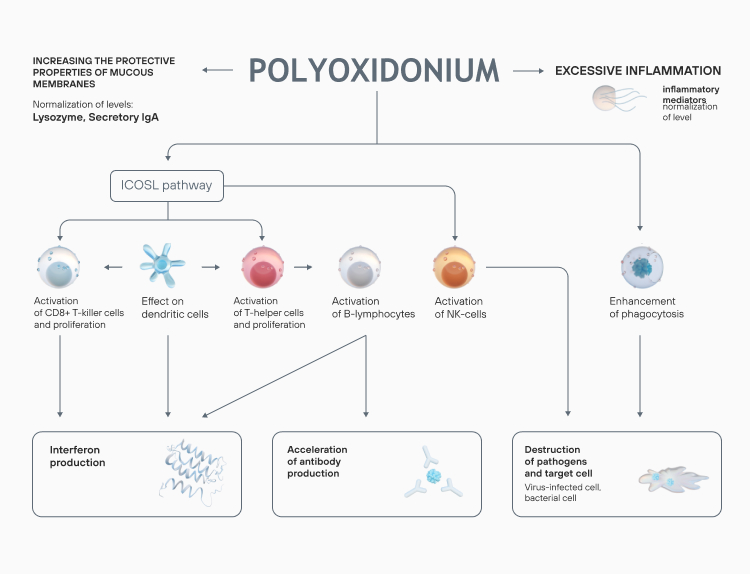
The drug is used to treat a wide range of diseases both on its own and as part of a complex therapy.
In 2019, a meta-analysis of controlled clinical trials evaluating the efficacy of azoximer bromide in the treatment of respiratory tract inflammatory infections in children and adolescents was conducted.


Meta-analysis of controlled clinical trials with respect to the use of azoximer bromide in the therapy of respiratory system inflammatory infections in children.
The results were demonstrated in another meta-analysis, which evaluated the effect of Polyoxidonium on the risk of acute respiratory infection and coronavirus in medical staff. 718 people took one tablet of the drug for 30–40 days, while 423 people made up the control group. Taking Polyoxidonium increased the odds of health professionals not getting ARI and COVID-19 by 3.02 times, and not getting COVID-19 alone — by 2.65 times.


Another review of the results of randomized, placebo-controlled, double-blind multi-center phase II and III clinical trials analyzed the results of azoximer bromide use for the treatment of influenza and ARVI in children aged 3 to 14 years (228 people).


During the coronavirus pandemic, studies were conducted on the efficacy of azoximer bromide in the therapy of the disease and post-COVID syndrome.
The possibility of including azoximer bromide in the therapy of coronavirus was confirmed by clinical trials. In 2022, the results of a study on the use of the drug in patients with COVID-19 (81 patients) were published.


The primary endpoint included clinical status on the 15th day compared to baseline. Data on 100 patients from a randomized controlled open-label study conducted in China were used as the control group.
The average length of hospital stay was comparable in the both groups. By the 28th day, the mortality rate of patients in the control group was 25%; none of the azoximer bromide group patients died during the study (one patient died on the 34th day after the end of the study).
The survival rate was statistically significantly higher in the azoximer bromide group than in the control group
| Mortality rate at day 28, n (%) | Azoximer bromide group, n = 77 | Historical control group, n = 100 |
|---|---|---|
| Alive | 77 (100,0) | 75 (75,0) |
| Died | 0 (0,0) | 25 (25,0) |
The patients’ state was assessed using a 7-point validated standardized scale. By day 14–15, in the control group the improvement was insignificant (3.87 points compared to 3.99 at the beginning of the treatment), in the drug group a significant improvement was observed: 2.90 points compared to 4.36 at the beginning of the therapy. Thus, the drug showed its important role as part of a complex therapy.
In another study, which evaluated the effect of azoximer bromide on the number of COVID-19 infections among healthcare professionals and on the level of chronic fatigue, the efficacy of the drug was also shown. There were 78 participants in the study (41 of them took azoximer bromide for 38 days, the comparison group had 37 participants).
The number of those who contracted COVID-19 in the experimental group was lower than in the comparison group. There was a statistically significant faster reduction in the manifestations of the chronic fatigue syndrome in the study participants who took azoximer bromide.
Effect of prophylactic azoximer bromide administration on the incidence of COVID-19 during the 68-day study period


The incidence of the SARS-CoV-2 infection was statistically significantly lower in study participants, which received azoximer bromide as a prophylactic drug, than in the comparison group.
These findings were confirmed in another independent study involving 913 healthcare professionals working in hospital ‘red zones’. The use of azoximer bromide reduced the number of ARVI and coronavirus infections, as well as reduced the risk of pneumonia.
In the study of the effectiveness of Polyoxidonium in the therapy of post-COVID syndrome, it was shown that after 10 days of taking the drug, joint and muscle pain, headache, olfactory disorders were noted less frequently, and dizziness became less pronounced. Already on the fifth day of the experiment, the patients of the experimental group showed significantly less general weakness compared to the control group.
Azoximer bromide is suitable for the treatment and prevention of influenza and ARVI in children, as well as for the treatment of acute and chronic infectious diseases of the ENT organs, upper and lower respiratory tract. For children from 6 months of age, Polyoxidonium in the form of solution can be administered intramuscularly, intravenously, intranasally or sublingually.
For children over 1 year old, the drug in the form of suppositories is suitable, and patients over 3 years old can be given the drug in the form of tablets.
Solution 6 mg/mL, parenterally.
Treatment of moderate and severe forms of
a disease, parenterally (intravenously and intramuscularly) 0.1 mg/kg for 3
consecutive days. Then every other day. Total course of treatment — 10 days.
Prepared solution for parenteral administration should not be stored.
In case of tenderness at the injection site, Polyoxidonium solution can be used in the same syringe with procaine (novocaine) if a patient has no hypersensitivity to it. For this purpose, 1 mL of 0.5% procaine (novocaine) solution is added when transferring the drug solution from a vial to a syringe.
Azoximer bromide strengthens the body’s response to infection at any stage of a disease, reduces the risk of viral, bacterial, and fungal infections. Polyoxidonium reduces the frequency of repeated ARVI: the results of a placebo-controlled, double-blind trial showed that the frequency of viral respiratory diseases in children, who underwent a 7-day treatment course, decreased 2-fold six months after the therapy.
In 2022, the results of an open-label, comparative, cohort study of the efficacy of Polyoxidonium as a means of preventing ARVI, influenza and coronavirus in children attending kindergartens and schools, were published. The children from the first and second groups (300 participants in each) received Polyoxidonium for 3 months and underwent 3 and 6 prophylactic courses, respectively. The children from the third control group (also 300 participants) did not receive the drug.
In the azoximer bromide groups, a significant reduction in the number of ARVI and COVID-19 infections was recorded. The drug reduced the risks of complications (pneumonia), increased nonspecific resistance of the upper respiratory tract mucosa.
As shown in a meta-analysis in 2019, the use of azoximer bromide also accelerates patient recovery and provides relief. Analysis of data from 540 people aged 3 to 18 years showed that: Polyoxidonium accelerates normalization of temperature, reduces the duration of symptoms of fever and intoxication, headache, muscle and joint pain, as well as the duration of clinical symptoms of acute inflammation of the upper respiratory tract.
Polyoxidonium is used as part of a complex therapy of ENT organs, it is included in the Standards of Medical Care in otorhinolaryngology.
Azoximer bromide in the treatment of acute and chronic infectious diseases of ENT organs can help normalize nasal breathing already by the 4th day of treatment, reduce the size of inflamed tonsils, 2 times reduce the duration of cough and running nose, normalize the microbial composition of the nasopharynx. The drug helps reduce the risk of repeated respiratory tract diseases and complications.
Another study, conducted with the participation of 110 children aged 3 to 7 years, showed that the use of Polyoxidonium in patients with hypertrophy of the palatine tonsils reduces the manifestation of nasal breathing disorders, reduces the frequency of ARVI and increases antimicrobial peptide gene expression.
Polyoxidonium is used for the treatment of pneumonia. It was shown that inclusion of azoximer bromide in the complex therapy of out-of-hospital pneumonia alleviates the course of the disease and shortens the period of intoxication.

Azoximer bromide can increase the efficacy of therapy of inflammatory infections of the genitourinary system and female genital organs, including:
The drug enhances the effect of anti-inflammatory drugs, accelerates tissue regeneration after surgery, reduces the period of manifestation of inflammatory processes and irritation of the mucous membrane, increases the body’s resistance to infections.